Introduction
Dental implant treatment is highly reliable and shows a favorable long-term prognosis (Buser 2012, Wittneben 2013). Studies have indicated that a buccal bone thickness of 1.5-2 mm following implant placement is essential for long-term success (Buser 2013, Monje 2019). This requirement is attributed to the progressive bone resorption around implants over time, directly linking initial bone volume to prognosis (Ramanauskaite 2022).
In cases where insufficient buccal bone exists after implant placement, a gingival recession might compromise the esthetic appearance and make proper oral hygiene maintenance difficult. Consequently, this increases the risk of peri-implant diseases (Schwarz 2018, Monje 2019).
Therefore, in cases of atrophic alveolar bone due to periodontal disease, guided bone regeneration (GBR) becomes necessary. GBR is a technique that achieves bone regeneration by applying bone graft materials and barrier membranes to bone defects (Dahlin 1988). The stability of the grafted bone is a critical factor for success in GBR. Membranes are available in both resorbable and non-resorbable forms. While resorbable membranes demonstrate superior biocompatibility to non-resorbable ones, they exhibit lower form stability. Conversely, non-resorbable membranes offer excellent form stability but require subsequent removal.
Studies have reported that stable bone augmentation can be achieved in horizontal bone defects using resorbable membranes secured with fixation pins (Mir-Mari 2015). However, research has also shown that bone resorption around the platform may occur even when combining resorbable membranes with fixation pins during simultaneous implant placement and GBR (Goran I Benic 2019). These findings indicate that graft material stability varies depending on bone defect morphology, necessitating appropriate barrier membrane selection for each specific situation. This article evaluates bone defect classification three-dimensionally and discusses the optimal choice of barrier membrane based on these morphological characteristics.
Classification of bone defect morphology
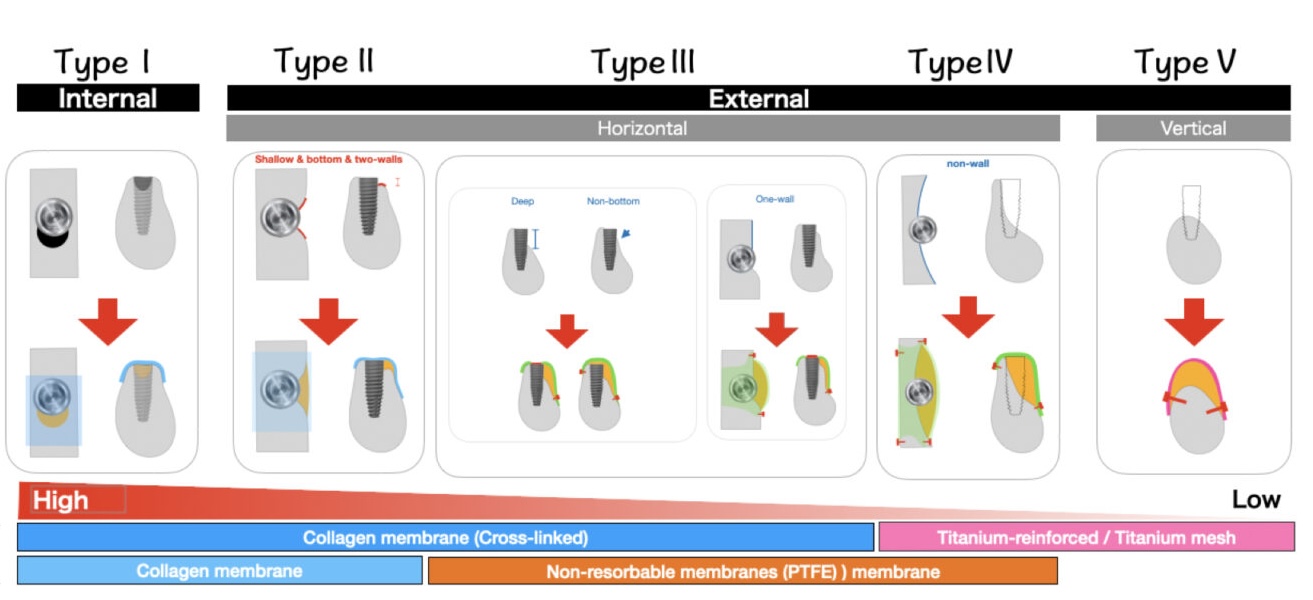
Bone defects in implant treatment can be classified into five types based on their morphology and depth. This proposed classification serves as an essential indicator when selecting treatment approaches (Figure 1). Types I-Ⅳ allow for simultaneous bone augmentation with implant placement when sufficient initial implant stability can be achieved. Conversely, Type V might require bone augmentation before implant placement. The characteristics of each type and appropriate membrane selection are detailed below.

Figure 1:
Type I: Intraosseous defects – These defects are contained within the alveolar bone. The addition of bone substitutes allows for highly stable space maintenance as alveolar bone walls surround the bone defect. In such cases, covering the defect with a collagen membrane effectively prevents soft tissue infiltration and yields favorable results. Due to the inherent mechanical stability afforded by the graft material, even collagen membranes without form stability can lead to sufficient bone regeneration.
Type II: Stable horizontal bone defects
- Characterized by shallow horizontal bone defects where less than 1/3 of the implant body is exposed
- Features a base of alveolar bone on the buccal side
- Demonstrates alveolar bone walls on both mesial and distal aspects
These conditions ensure graft stability. As with Type I, covering the bone graft with a collagen membrane produces favorable outcomes. The use of resorbable fixation pins or sutures can provide enhanced stability.
Type III: Unstable horizontal bone defects –These defects lack one or more of the three elements of Type II horizontal bone defects. Due to decreased graft stability, non-resorbable membranes such as PTFE or titanium membranes are recommended. When using a collagen membrane, a cross-linked collagen membrane with delayed resorption is recommended. These membranes require secure fixation with pins or sutures.
Type IV: Severe horizontal bone defects –These cases present severely atrophic alveolar bone where more than 2/3 of the implant body would be exposed. When initial implant stability cannot be achieved due to the extensive bone defect and unstable graft material, a two-stage approach is recommended, performing bone augmentation before implant placement. Due to graft instability, using titanium-reinforced PTFE membranes or titanium membranes with secure fixation using pins is recommended.
Type V: Vertical bone defects – These defects require vertical bone augmentation. Use of highly form-stable titanium membranes, titanium-reinforced PTFE membranes, or titanium mesh is recommended. Due to the extensive nature of the bone defects, secure fixation with pins and adequate soft tissue coverage are essential.
*Note 1: These classifications assume standard-length implants (8-10 mm).
*Note 2: Although this article focuses on membranes, some techniques do not require them, such as those using autogenous bone blocks and fixation screws.
Barrier membrane characteristics and selection criteria with clinical cases
The various barrier membranes used in GBR for implant treatment can be classified into three major categories based on their characteristics. Each membrane type exhibits unique properties and should be selected appropriately for bone defect morphology.
Native Collagen Membranes
Structure and properties
- Composed primarily of Type I collagen
- Demonstrates high biocompatibility and resorbability
- Typically undergoes resorption within approximately 2 months, eliminating the need for secondary removal surgery
- Exhibits excellent soft tissue compatibility and promotes favorable wound healing
Features and functions
- Enhanced initial soft tissue healing due to superior biocompatibility
Suitable applications
- Particularly effective for Type I and II defects
- Optimal for cases where bone walls provide support and graft material stability
Precautions
- Limited form stability and rapid resorption make it unsuitable for unstable graft cases
- Accelerated resorption occurs if membrane exposure occurs
CASE 1 (Type Ⅰ)
The intraosseous defect was observed due to impaired healing of the extraction socket. After placing bone graft material, the site was covered with a resorbable collagen membrane (Bio-Gide ® Geistlich). At second-stage surgery, successful bone regeneration was achieved.
Cross-Linked Collagen Membranes
Structure and properties
- Composed of Type I collagen that has undergone cross-linking treatment
- Features an extended resorption period of 4-6 months compared to conventional collagen membranes
Features and functions
- Demonstrates resistance to tissue enzymatic degradation while maintaining cellular compatibility
Suitable applications
- Effective for Type I, II, and III bone defects
Precautions
- Higher risk of exposure-related complications compared to non-cross-linked membranes
- If exposed, requires intervention due to slower degradation than standard collagen membranes
CASE 2 (Type Ⅱ)
After implant placement, the buccal bone base was present with two-wall bone support in the mesial and distal aspects. Due to the stability of the bone graft material, a cross-linked collagen membrane (Ossix ® Plus) was applied with suture. After approximately 6 months of healing, sufficient bone regeneration was observed.
D-PTFE Membranes
Structure and properties
- Features high-density PTFE providing reliable barrier function
- Non-resorbable with excellent strength and form maintenance
- Allows secure fixation with titanium pins or fixation screws
- Titanium-reinforced versions offer enhanced form stability
Features and functions
- Demonstrates superior barrier function against bacterial invasion
- Enables long-term maintenance of form
Suitable applications
- Particularly effective for Type III, IV, and V defects
- Indicated for cases requiring extensive bone augmentation
- Titanium-reinforced versions specifically suitable for stabilizing large defect areas
Precautions
- Requires secondary surgery for removal
- The risk of membrane displacement or exposure increases without proper fixation
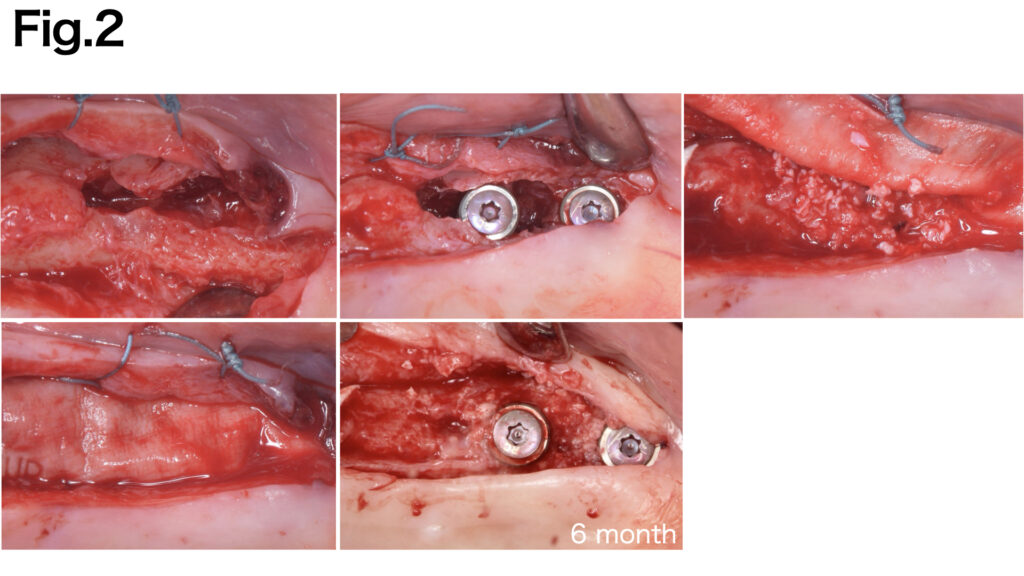
CASE 3 (Type Ⅲ)
Despite minimal exposure to the rough surface, the case presented the absence of both buccal bone base and mesial-distal bone walls. A non-resorbable d-PTFE membrane (CytoplastTM TXT-200) was applied to stabilize the grafted bone. The buccal aspect of the membrane was secured with fixation pins, while the superior portion was fixed with the implant cover screw. Adequate bone regeneration was achieved following the healing period.
E-PTFE Membranes
Structure and properties
- Composed of expanded PTFE featuring microscopic pores with enhanced flexibility
- Non-resorbable, ensuring stability throughout the bone-regeneration period
- Presents potential risk for bacterial infiltration
- Titanium-reinforced versions demonstrate improved form maintenance capability for extensive defects
Features and functions
- Exhibits superior space maintenance capability for bone formation
- Requires primary closure to minimize infection risk
- Promotes controlled tissue regeneration
Suitable applications
- Particularly effective for Type III, IV, and V defects
- Indicated for cases requiring extensive bone augmentation
- Titanium-reinforced versions specifically suitable for stabilizing large defect areas
Precautions
- High risk of bacterial infection upon exposure
- Essential to maintain proper soft tissue management
- Not recommended in areas where primary closure is challenging
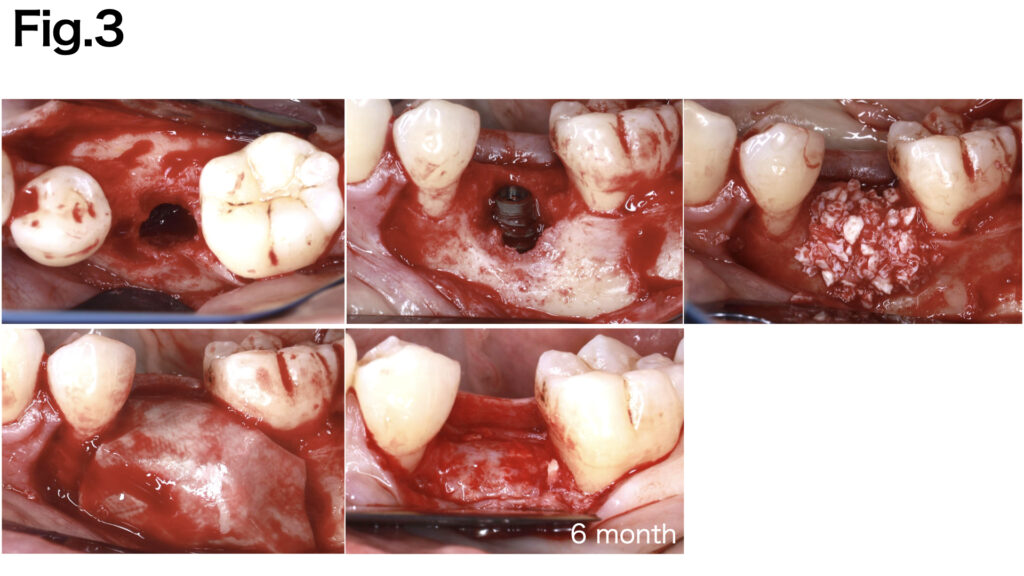
CASE 4 (Type Ⅲ)
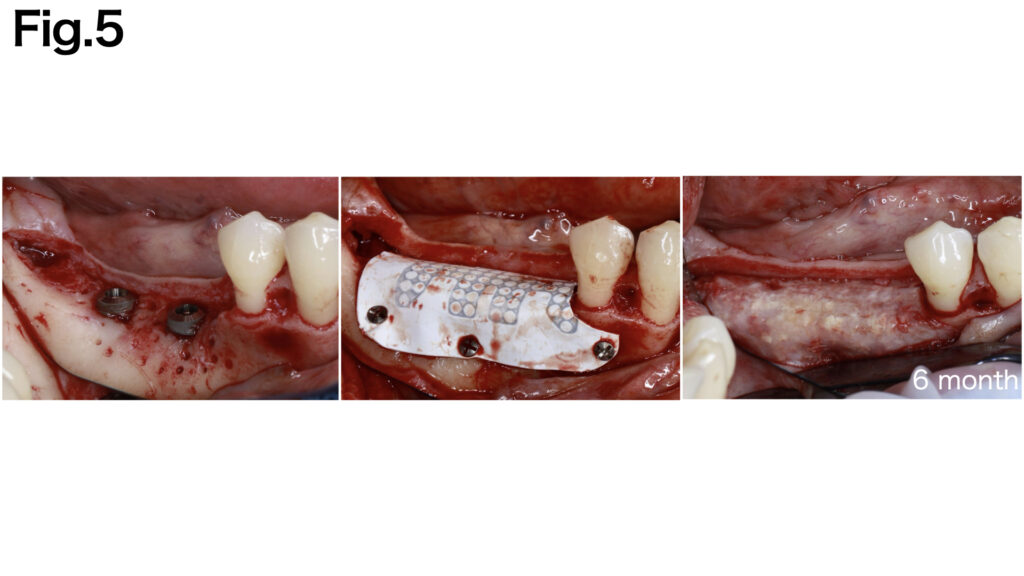
Due to the extensive width of the bone defect and the absence of basal bone, stabilizing the grafted bone was challenging. Fixation pins were applied to both the buccal and lingual aspects to achieve secure fixation of the e-PTFE membrane (NeoGen®). After six months of healing, sufficient bone regeneration was achieved.
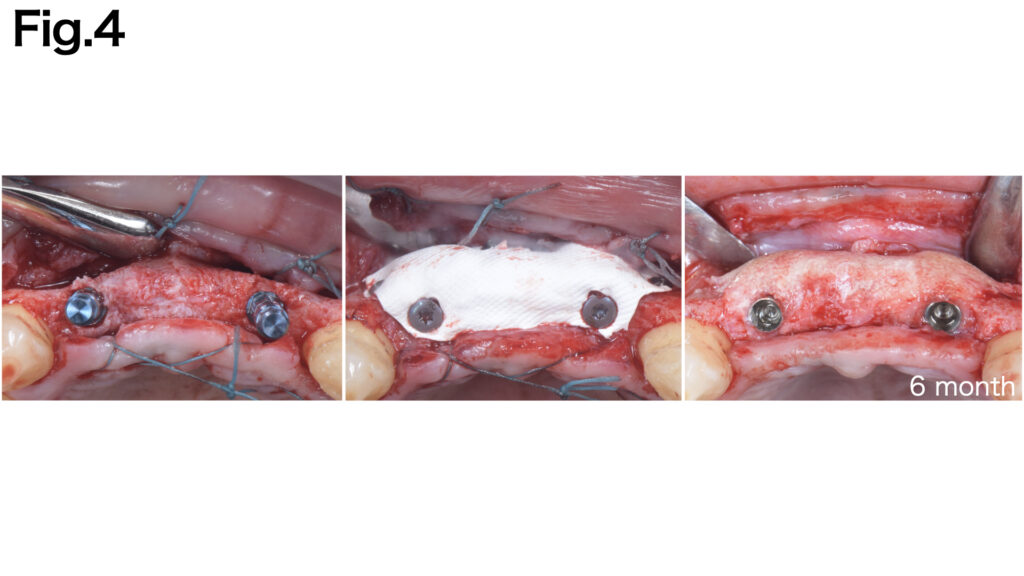
CASE 5 (Type Ⅳ)
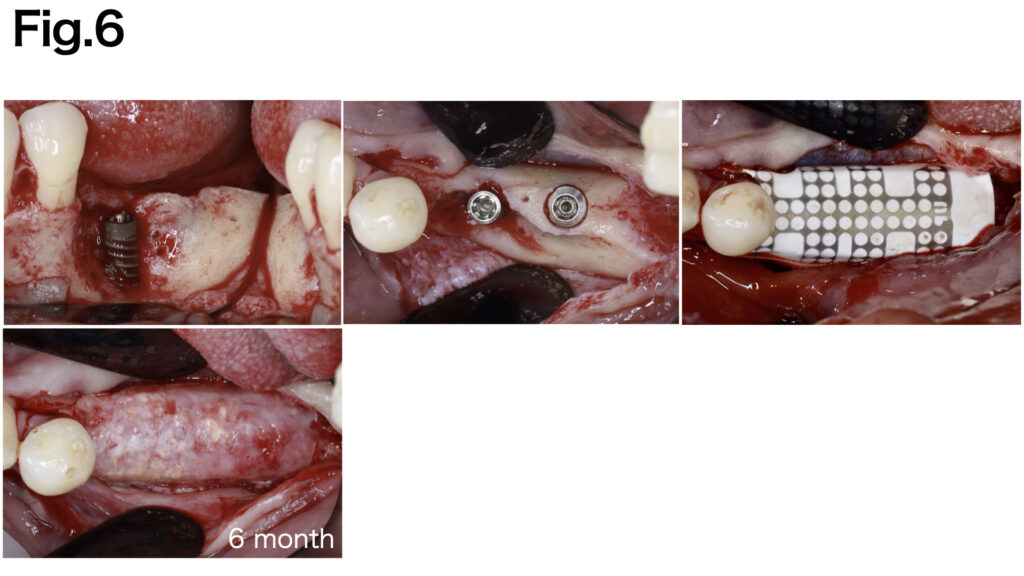
A severely atrophic horizontal bone defect was observed. Fixation pins were applied to both the buccal and lingual aspects to achieve secure fixation of the e-PTFE membrane (NeoGen®︎). After six months of healing, adequate bone regeneration was achieved.
Titanium Membrane
Structure and properties
- Manufactured from pure titanium with a high-strength design
- Demonstrates excellent form stability and shape maintenance
- Enables secure fixation through screw placement
Features and functions
- Superior formability allows ideal bone contour restoration even with significant defects
- Provides stable graft material containment through rigid fixation
Suitable applications
- Optimal for Type III, IV and V defects
- Achieves predictable bone regeneration in extensive defect cases
- It is particularly effective where mechanical strength is essential
Precautions
- Necessitates secondary surgery for removal
- Increased infection risk upon exposure
- Complete soft tissue coverage is crucial
CASE 6 (Type V)
The vertical bone defect was identified. Fixation pins were applied to both the buccal and lingual aspects to achieve secure fixation of the titanium membrane (Ti-honeycomb membrane, TiHM). After six months of healing, adequate bone regeneration was achieved.
Titanium Mesh
Structure and properties
- Features high-form stability through a pure titanium mesh structure
Features and functions
- Promotes blood flow maintenance
- Adaptable to complex three-dimensional bone defects
- Enables secure fixation when combined with titanium screws
- Can be customized using CAD-CAM technology to match the specific defect
Suitable applications
- Particularly indicated for Type V (vertical bone defects)
- Achieves predictable outcomes in extensive vertical bone augmentation cases
- Ideal for complex three-dimensional defects
Precautions
- Requires collagen membrane coverage to prevent mesh exposure and soft tissue invasion into the mesh
- Necessitates secondary surgery for removal
- High risk of infection upon exposure
- Risk of tissue damage during removal due to soft tissue integration into mesh structure
CASE 5 (Type Ⅴ)
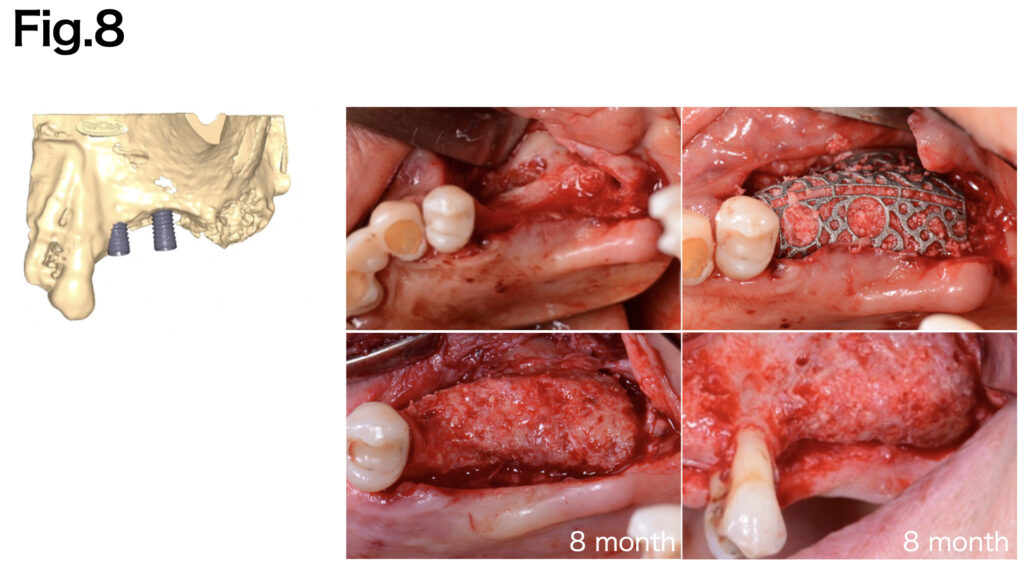
The vertical bone defect was identified. Fixation pins were applied to achieve secure fixation of the titanium mesh (Yxoss CBR® (ReOSS)). After six months of healing, adequate bone regeneration was achieved.
Summary and discussion
In bone augmentation procedures for implant treatment, proper evaluation of bone defect morphology and appropriate barrier membrane selection significantly influence treatment predictability. The bone defect classification system (Types I-V) presented in this article has been organized from the perspective of graft material stability, providing specific criteria for membrane selection in various clinical situations.
Resorbable collagen membranes demonstrate sufficient therapeutic efficacy for cases with simple bone defect morphology and stable graft materials (Types I-Ⅲ) (Case 1, 2). Conversely, unstable horizontal bone defects (Type III) require non-resorbable membranes such as PTFE or titanium (Case 3). Furthermore, more severe horizontal bone defects (Type IV) necessitate the application of titanium-reinforced PTFE membranes or titanium membranes (Case 4). For vertical bone defects (Type V), secure form maintenance and fixation using titanium mesh or titanium-reinforced PTFE membranes become essential (Case 5). The successful outcome of bone regeneration procedures depends on several key factors:
- Accurate assessment of the bone defect morphology
- Selection of appropriate barrier membranes based on bone defect morphology
- Proper fixation techniques
- Adequate soft tissue coverage through sufficient releasing incisions to prevent membrane exposure
The clinician must consider these factors comprehensively when planning and executing bone augmentation procedures. Additionally, it is crucial to understand that each membrane type has its specific advantages and limitations, making case-specific selection essential for optimal outcomes.
Based on bone defect morphology, this systematic approach to barrier membrane selection provides clinicians with a practical framework for achieving predictable outcomes in guided bone regeneration procedures.